Current Oncology, Free Full-Text
Por um escritor misterioso
Descrição
Background: Information on immune responses in cancer patients following mRNA COVID-19 vaccines is still insufficient, but generally, patients had impaired serological responses, especially those with hematological malignancies. We evaluated serological response to COVID-19 mRNA vaccine in cancer patients receiving chemotherapy compared with healthy controls. Methods: In total, 195 cancer patients and 400 randomly selected controls who had been administered a Pfizer-BioNTech or Moderna COVID-19 vaccines in two doses were compared. The threshold of positivity was 4.33 BAU/mL. Patients were receiving anticancer treatment after the first and second dose of the vaccines. Results: a TOTAL OF 169 patients (87%) had solid tumors and 26 hemolymphopoietic diseases. Seropositivity rate was lower in patients than controls (91% vs. 96%), with an age/gender-adjusted rate ratio (RR) of 0.95 (95% CL = 0.89–1.02). Positivity was found in 97% of solid cancers and in 50% of hemolymphopoietic tumors. Both advanced and adjuvant therapy seemed to slightly reduce seropositivity rates in patients when compared to controls (RR = 0.97, 95% CL = 0.89–1.06; RR = 0.94, 95% CL = 0.87–1.01). Conclusions: the response to vaccination is similar in patients affected by solid tumors to controls. On the contrary, hemolymphopietic patients show a much lower response than controls.
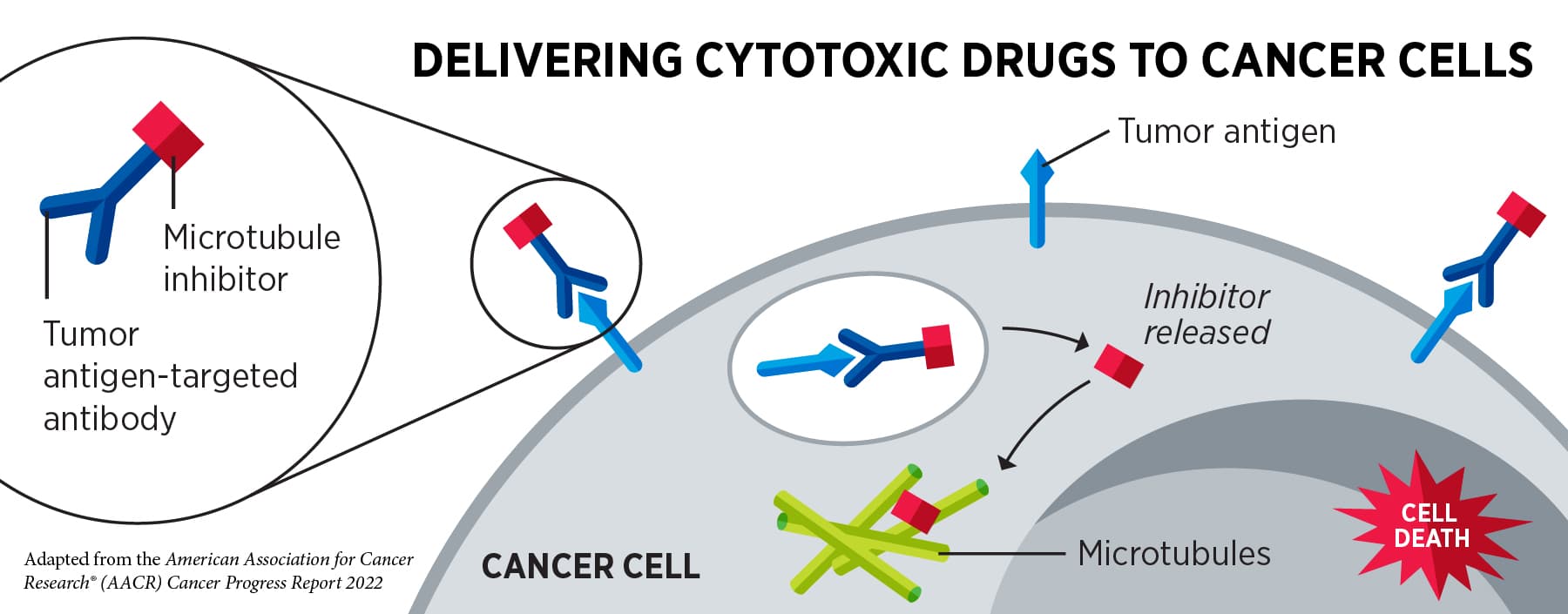
FDA Approvals in Oncology: April-June 2023 - American Association for Cancer Research (AACR)
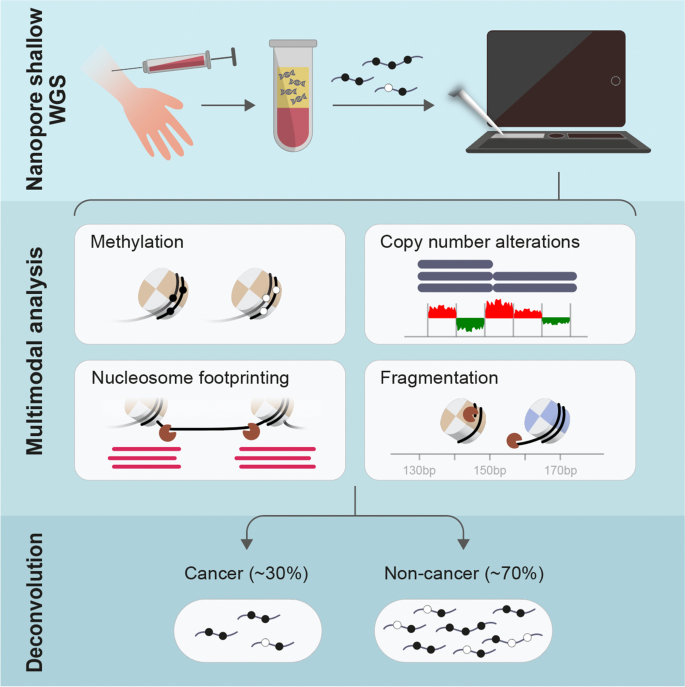
Detecting cell-of-origin and cancer-specific methylation features of cell- free DNA from Nanopore sequencing, Genome Biology

Malignant: How Bad Policy and Bad by Prasad, Vinayak K.

Journal of Current Oncology and Medical Sciences

Current Oncology An Open Access Journal from MDPI

Download or order free information booklets
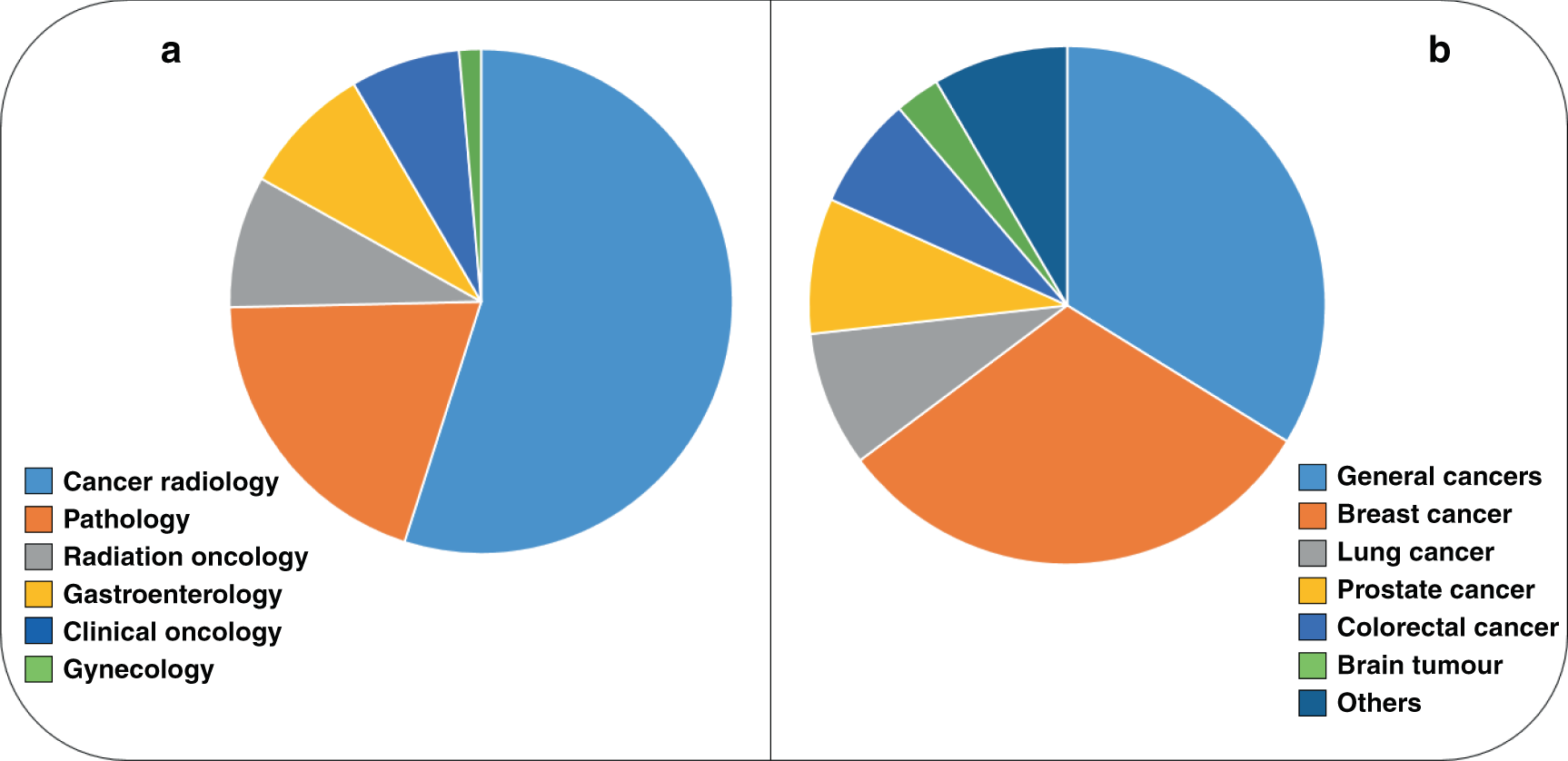
Artificial intelligence in oncology: current applications and future perspectives
Asia-Pacific Journal of Clinical Oncology - Wiley Online Library

PDF) Secular Trends in the Survival of Patients with Laryngeal Carcinoma, 1995–2007
de
por adulto (o preço varia de acordo com o tamanho do grupo)






